How Penile Vibratory Stimulation helps men recover erectile functioning following Prostate Cancer Treatment?
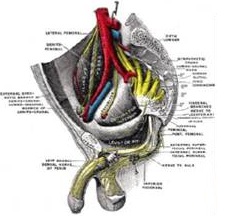 Penile erections are nerve generated events that cause blood to fill the penis and several mechanisms preventing the blood from leaking out of the penis. Unfortunately, after prostate cancer treatment most men will develop defective erectile mechanisms like:
Penile erections are nerve generated events that cause blood to fill the penis and several mechanisms preventing the blood from leaking out of the penis. Unfortunately, after prostate cancer treatment most men will develop defective erectile mechanisms like:
- Poor nerve conduction to the penis
- Poor blood flow to the penis
- Development of venous leakage
Current treatments such as oral medications and injections do not address all of these problems. Surgery and Radiation cause damage to the Cavernous Nerves (nerves that help facilitate penile erection) which are very close to The Prostate. Nerve conduction is reduced or stopped altogether for a variable amount of time. Cavernous Nerves are (neural) highways to take information from the spinal cord to the penis.



